In part one I gave a basic overview of autophagy and if you missed that blog you can read it here.
Before I jump into part two, let me provide a quick recap of part one. We learned that autophagy is where cell components are sequestered and eaten. We learned that autophagy happens all the time (to a degree) but it increases under stressful conditions like nutrient deprivation, and so fasting will boost it. We learned that autophagy is different from apoptosis, which is programmed cell death. We also learned that autophagy can be driven by supply or demand. Autophagy by demand occurs as a response to a stressor like nutrient deprivation, autophagy by supply occurs when a damaged or aging component is targeted for breakdown because it’s giving signals to be eaten. Lastly, we learned four key steps of macroautophagy.
I also introduced two autophagy heroes; Toffee, the autophagy aardvark, and the Apoptosis Assassin.
In part one I focused on what autophagy is, but what actually causes autophagy? I’ve already said that we can induce it through fasting. But how? Well, it has to do with nutrient deprivation and pathways involved in nutrient sensing. There seem to be three key pathways that drive autophagy. One is called protein deacetylation, one is called mTOR, and one called AMP Kinase. All these pathways are in some shape or form nutrient and or energy sensing. Let’s talk about them.
The first pathway that triggers autophagy is the mTOR pathway. Or to be more specific, the blocking of this pathway. It’s the inhibition of the mTOR that is a driver of autophagy.
What is mTOR? It stands for the Mechanistic Target of Rapamycin and its activation is associated to building. It’s actually a protein kinase that regulates growth, cell proliferation and protein synthesis (along with quite a few others as well). It’s an ancient pathway and gets some of its cues by sensing the nutrient, oxygen and energy levels within cells.
So if mTOR inhibition drives autophagy, the obvious question is, how do we inhibit it?
To answer that, we first need to understand what activates it. It turns out nutrients will activate it, but in particular amino acids, especially one called leucine, a branched chain amino acid. Amino acids are what proteins are made of, some proteins are made up of 100s, some are made up of 1000s.
So, if we eat protein, we are essentially eating amino acids and so we activate mTOR. If we don’t eat protein, but we still eat carbs and fat, we’ll dampen mTOR somewhat. However, if we don’t eat ANYTHING i.e. we fast, we’ll dampen it even more and we’ll get that some of that inhibition we’re looking for. Remember, mTOR is energy and nutrient sensing. So, no food, no nutrients, no energy, no mTOR activation and so we have autophagy.
Now, to help remember the mTOR pathway, I’d like to introduce another autophagy hero. This one is called Thor mTOR. Thor mTOR is all about growth, cell proliferation, protein synthesis and big muscles. If you give Thor MTOR some protein, he is happy and activated and will use his powers to ensure his environment is growing.

When Thor is fed and activated, he does NOT like Toffee; the autophagy aardvark. They don’t work well together at all. i.e you can’t really have autophahgy when MTOR is activated, as we have aleady established.

If Toffee wants to sneak in and do his autophagy thing, then Thor has to be starving weak or asleep (i.e. inhibited or not activated). Now, there are of course exceptions but for now let’s go with that as a general rule to help us remember this a little easier.

So, that’s the first trigger of autophagy; the inhibition of the mTOR pathway.
The second trigger of autophagy is the AMP-Kinase pathway, or to be more specific, the activation of the AMP-Kinase pathway.
What is AMP Kinase? Well, it’s another protein Kinase and it stands for adenosine monophosphate-activated protein kinase. AMP Kinase is all about balance and regulating cellular energy homeostasis. Like mTOR, it is also senses and gets cues from cellular energy levels. But when energy is low, AMPK is activated.
When activated, AMPK does many things, including blocking the synthesis of triglycerides (or let’s call it fat), as well as activating beta oxidation (the breaking down of fat in the liver) and activating ketogenesis (the formation of ketones which are like a form of fuel). Now, doing all these things is consistent with the idea of AMP Kinase regulating cellular energy homeostasis. When energy is low, we don’t want to be creating fat stores, we want to be making real time energy! So, AMP Kinase is activated to block fat synthesis and activate the breaking down of fat for fuel.
So the activation of autophagy through the activation of AMP Kinase again ties back to fasting. When we don’t eat, our energy levels are low and the ATP (adenosine triphosphate) level of our cells goes down. ATP is like the energy currency of cells. When ATP levels are low, AMP Kinase is activated and we get autophagy.
Now, to help remember this pathway, let’s enter another autophagy hero. This one is called the AMP Kinase King, or just the Kinase King for short. The Kinase King is kind of a hippy king, he’s spiritual and he’s all about energy and keeping balance in the Universe.

The Kinase King gets his cues form cellular energy levels. So, he is very watchful of the ‘ATP globe’. When cellular energy or ATP levels are high, the Kinase King can relax and take it easy (i.e. is not activated).

However, when the ATP levels are low, that’s when he gets activated and springs into action to restore energy homeostasis.

He blocks triglyceride or fat formation from fatty acids, because that’s not what we want to be doing when there is not much energy…..

We want to be creating energy when energy is low, so the Kinase King will trigger beta oxidation and ketogenesis which both break down fatty acids to form energy ..

So, that’s the second major activator of autophagy, the activation of AMP Kinase.
Now let’s talk about the third one, which is protein deacetylation –
Before we talk about how autophagy is triggered by protein deacetylation, we need to establish what the protein deacetylation pathway is. And the best way to do that, is to first talk about protein acetylation.
What is protein acetylation?
At the highest level, you could say it is when the acetyl group of an acetyl-CoA molecule, is added to a protein. That process is called post translation modification, or PTM.
Why does PTM occur? Well, the purpose of this process is to change the function of the protein. It’s the step that sometimes occurs following the creation of a protein. Taking things right back, we first start with DNA, and through a process called transcription, we create the RNA. Then from the RNA, through a process called translation, we create the protein. Finally, the protein may undergo post translational modification to alter its function.
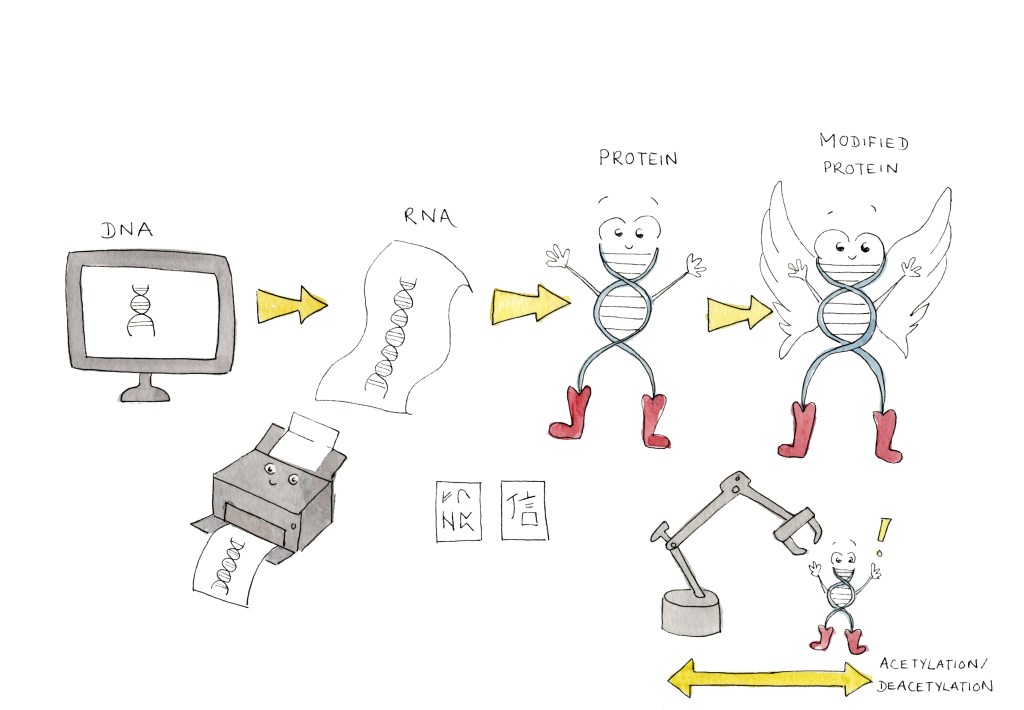
So now we know what protein acetylation is and why it occurs, let’s talk about how it occurs. I said that the acetyl group of an acetyl-CoA molecule is added to a protein. This is done via an enzyme called an acetyl transferase, which, as the name implies, transports the acetyl-group to the protein. More specifically, it transports it to a specific amino acid that makes up the protein, as proteins are formed of many amino acids.
So, protein acetylation is where the acetyl group of an acetyl-CoA molecule is added to an amino acid by an acetyl transferase enzyme, that’s protein acetylation. BUT, I said that autophagy is triggered by protein deacetylation. So what is protein deacetylation?
Well, as you might of guessed, it’s pretty much the opposite of protein acetylation. Instead of adding the acetyl group to the protein, it is being taken away. Taking away the acetyl group still counts as a post translational modification to alter its function, but instead of the acetyl transferase delivering it, it is done by an enzyme called a deacetylase. An important group of deacetylases that do this, are called sirtuins, which are often referred to as longevity proteins.
Now, protein acetylation and deacetylation occurs on numerous amino acids, but they are believed to occur most commonly on lysine residues. Lysine is just a particular amino acid, and the residue of the amino acid is the part of the amino acid that makes it unique from other amino acids. So if you have a bunch of different amino acids and take away all the residues, and they will look identical
Amino acids with residues –

Amino acids without residues (identical) –
Now we know what protein deacetylation is (basically removing the acetyl group from a lysine residue by a deacetylase) and we know protein deacetylation triggers autophagy, let’s talk about what triggers deacetylation.
Well, it has to do with the levels of acetyl-CoA that are present in cytosol of the cell (remember, acetyl-CoA is the key molecule in the deacetylation process). The cytosol is basically the liquid inside a cell.
When we have high levels of acetyl-CoA, we get protein acetylation (I picture there being too many acetyl-COAs, so the number is being reduced by delivering to them to amino acids).
When the levels of acetyl-CoA are low, we get protein deacetylation (I picture there being not enough acetyl-CoAs, so the number is restored by pinching them from amino acids).
So, acetyl-CoA levels are low, we get protein deacetylation, we get autophagy. When do we have low levels of acetyl-CoA? To answer that, let’s look at how acetyl-CoA is created! I’m going to talk about three ways –
The Gylcolytic Pathway
Amino acid catabolism
Beta Oxidation
The glycolytic pathway is the breaking down of carbs or sugar. The amino acid catabolism is the breaking down of proteins. The beta oxidation is the breaking down of fat. Now, you will notice that these are the macronutrients that we find in food. So when we eat protein, carbs or fat, we make acetyl-CoA.
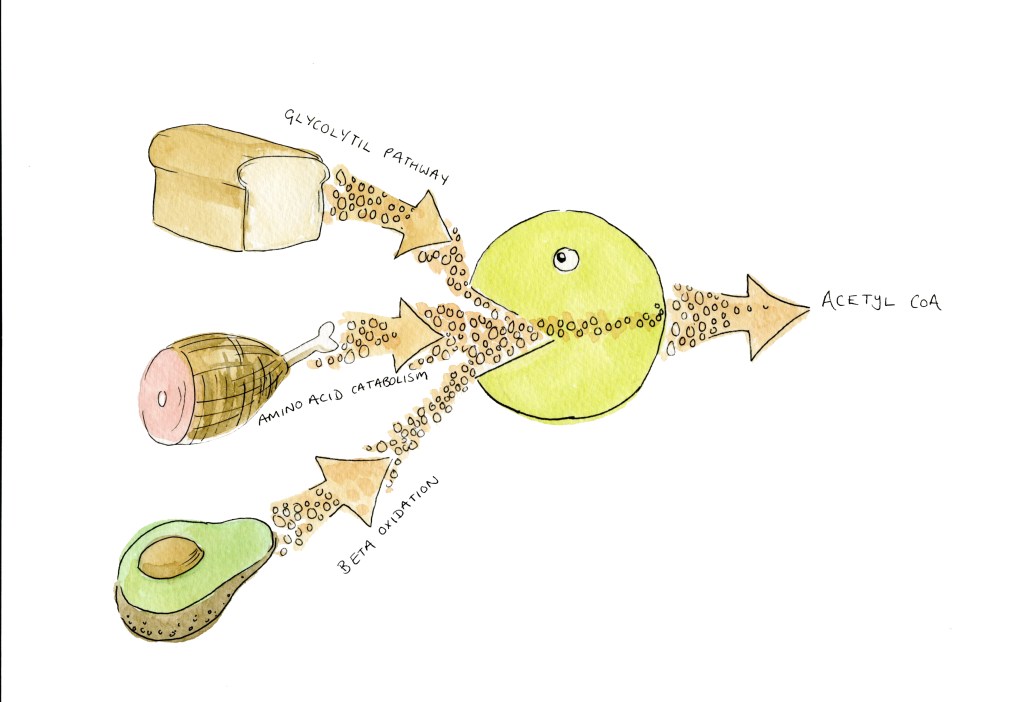
If we make lot’s of acetyl-CoA, the pool is high, and we get protein acetylation.
Now, if we take away protein, carbs and fat, i.e. we don’t eat or we fast, we don’t make Acetyl-CoA, our levels of acetyl-CoA are low, we get protein deacetylation, and we get……..autophagy.
Okay, so now its time to bring in some more character to help us remember this. The first one is actually a group of characters called the CoA Commandos. As you might of guessed, they represent the acetyl-CoA molecules.

The CoA commandos mean business, they are tough guys, military guys, and you could argue their main job is to enter the Krebs cycle and help to produce the energy of the body, ATP.
As I said, the commandos are tough guys, and such they like to hang around with other tough guys, so you can imagine they are good mates with THOR MTOR. They like to drink together and get rowdy. When lot’s of Commandos are in town, THOR is really happy and feels strong. They can be quite loud and boisterous, and some people are afraid of them.

The commandos (when lot’s of them are around and the CoA pool is high) feed into the acetylation process by working with the acetyl transferases by giving up their acetyl group, so the transferase can attached it to the protein…..
One set of characters in particular who really don’t like to be around when the CoA commandos are around, are the deer sitters, who represent the deacetylase enzymes. They are like dog sitters, but instead of looking after dogs, they look after deers…..

Remember, the deacetylases are responsible for removing the acetyl group from the lysine residue, which triggers autophagy, but they don’t like to do that while there are too many loud Commandos around, you know, they wouldn’t want to scare off the deer.
So when there aren’t that many CoA Commandos in town, i.e. the pool of Acetyl-CoA is low, the deer sitters come out, the deacetylase process takes place, and Toffee shows his face, and autophagy is triggered.
So! Those are the three pathways to activate autophagy! Thank you for reading to the end of the post, great job! Watch out for part 3 of the series, where I will talk about some different ways these three pathways can be triggered!